Management and monitoring are essential elements of the treatment journey
After treatment, patients need to continue to receive daily oral corticosteroids and be monitored on a regular basis for any changes in liver function and for other critical laboratory parameters.1
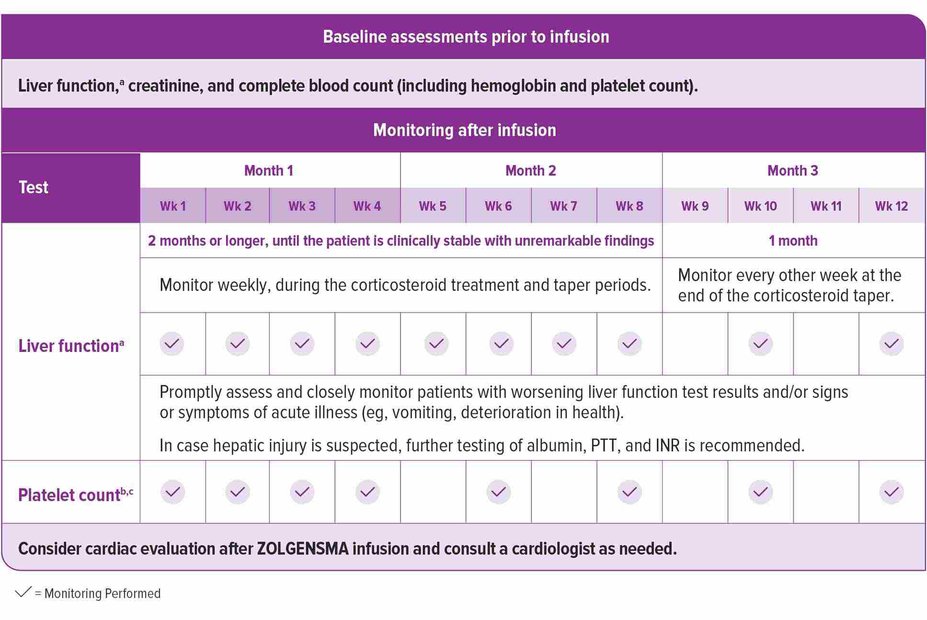
Laboratory testing before and after infusion
Prior to infusion, ensure patients have baseline tests for liver function,a creatinine, and complete blood count (including hemoglobin and platelet count). After infusion, continue to monitor liver function and platelet countb; additionally, consider conducting a cardiac evaluation if needed, as described in the Prescribing Information. See the recommended schedule of assessments in the accompanying chart.1
Due to the increased risk of serious systemic immune response, administer ZOLGENSMA to patients who are clinically stable in their overall baseline health status (eg, hydration and nutritional status, absence of infection) prior to infusion. Postpone ZOLGENSMA in patients with infections until the infection has resolved and the patient is clinically stable. Clinical signs or symptoms of infection should not be evident at the time of ZOLGENSMA infusion.1
Wk, week.
aLiver function assessment includes a clinical exam and laboratory testing of hepatic aminotransferases (aspartate aminotransferase [AST] and alanine aminotransferase [ALT]), total bilirubin, albumin, prothrombin time, partial thromboplastin time (PTT), and international normalized ratio (INR). Monitor liver function for 3 months or longer until results are unremarkable (normal clinical exam, total bilirubin, prothrombin, and INR results, and ALT and AST levels below 2 × ULN).
bMonitor platelet counts as well as signs and symptoms of thrombotic microangiopathy (TMA), such as hypertension, increased bruising, seizures, or decreased urine output.
cMonitor platelet counts for 3 months or longer until they return to baseline.
Treat with systemic corticosteroids
To help manage a possible increase in liver aminotransferases, treat all patients with systemic corticosteroids before ZOLGENSMA infusion and continue regimen after ZOLGENSMA infusion. If oral corticosteroid therapy is not tolerated, consider intravenous corticosteroids as clinically indicated.1
Day 1: 24 hours prior to infusion
Initiate systemic corticosteroid regimen equivalent to oral prednisolone at 1 mg/kg/day.
Day 2: Infusion day
Infuse ZOLGENSMA and continue the corticosteroid regimen.
Day 30: 28 days after infusion
Check liver status clinically and by assessing ALT, AST, total bilirubin, prothrombin time, and INR.
Do not stop systemic corticosteroids abruptly.
If unremarkable (normal clinical exam, total bilirubin, prothrombin time, and INR; ALT and AST levels below 2 x ULN):
Then taper the corticosteroid dose gradually over the next 28 days
If liver function abnormalities are present:
Consult a pediatric gastroenterologist or hepatologist
Continue systemic corticosteroids (equivalent to oral prednisolone at 1 mg/kg/day) until AST and ALT values are both below 2 × ULN and all other assessments return to normal range
Then taper the corticosteroid dose gradually over the next 28 days or longer if needed
30-day monitor following steroid taper
Once the patient is clinically stable with unremarkable findings at the end of the taper period, continue to monitor liver function every other week for another month.
ALT, alanine aminotransferase; AST, aspartate aminotransferase; INR, international normalized ratio; ULN, upper limit of normal.